Early detection of Alzheimer’s disease is no longer a matter of guesswork. Clinicians now marshal a broader set of tools to spot the earliest, most subtle changes, enabling timelier and more personalized care. While memory loss is not the only clue, an expanded diagnostic “toolbox” is making those first signs clearer and more reliable than ever.
A sharper clinical starting point
Evaluation still begins with the patient’s or family’s concerns, followed by structured neuropsychological testing. These assessments map attention, memory, language, executive skills, and daily functioning to confirm that true cognitive impairment exists. A careful clinical profile can already suggest a likely cause, but overlapping symptoms with other disorders demand greater specificity.
“It’s no longer enough to label changes as ‘normal aging.’ The goal is to define the pattern, the severity, and the likely pathology behind it.”
In parallel, clinicians screen for depression, sleep disorders, medications, thyroid or vitamin deficits, and sensory issues that can mimic or magnify cognitive complaints. This holistic view reduces misdiagnosis and anchors next steps in evidence.
Imaging that rules in—and rules out
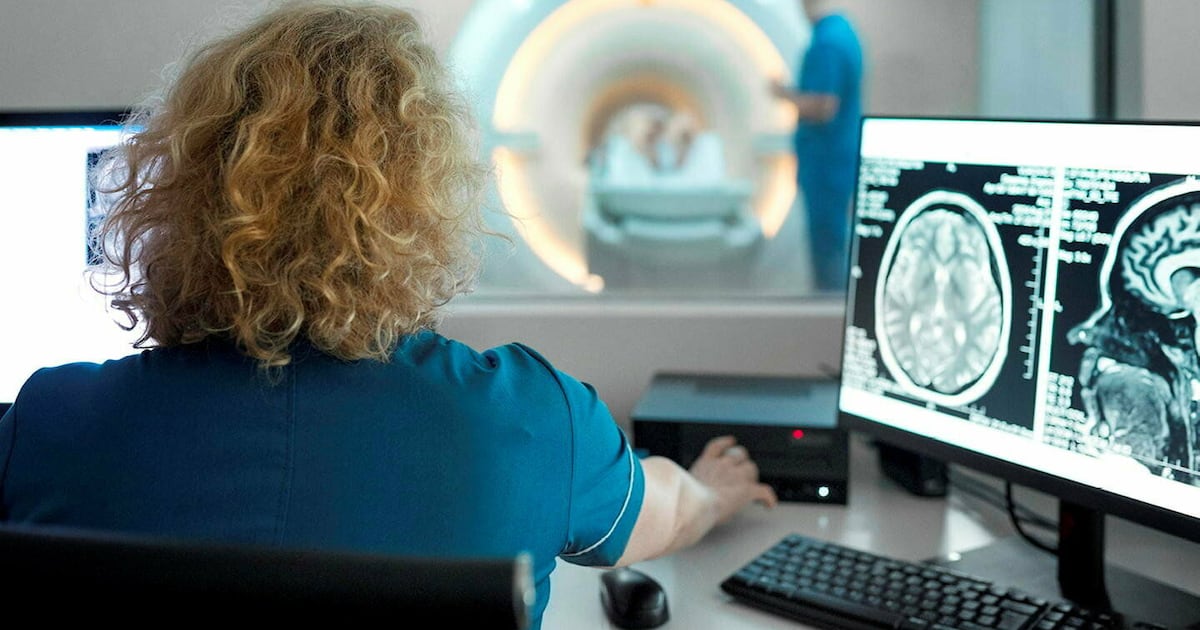
Neuroimaging has become indispensable for grounding the diagnosis. Brain MRI, the workhorse of clinical practice, helps exclude tumors, hydrocephalus, and large strokes while illuminating small-vessel disease that can drive vascular cognitive impairment. Early Alzheimer’s often involves the hippocampus; measurable hippocampal atrophy can support the hypothesis, though it is not specific and appears in other neurodegenerative conditions.
Where uncertainty remains, FDG- or amyloid-PET can refine the picture. These scans visualize metabolism or pathological plaques, respectively, tightening the diagnostic net when clinical and MRI findings are ambiguous. Because of cost and access constraints, PET is reserved for atypical, early-onset, or diagnostically challenging cases.
Biomarkers come of age
The most powerful advances center on biological markers of Alzheimer’s pathology. Cerebrospinal fluid testing can detect decreased Aβ42 (reflecting amyloid deposition) and elevated phosphorylated tau (reflecting tangle pathology and neurodegeneration) with high sensitivity and specificity. Together with markers of neuronal injury, these profiles build a near-definitive signature of disease.
Blood-based assays are quickly reshaping the landscape. Plasma phosphorylated tau (p-tau) and related panels now offer clinically available, minimally invasive options that correlate strongly with CSF and PET findings. While local availability and standards vary, blood tests are poised to widen early access and reduce reliance on lumbar puncture.
What early signs really look like
Beyond forgetfulness, the first changes often touch several cognitive and behavioral domains. Recognizing this broader spectrum helps families seek help earlier and clinicians triage more precisely:
- Subtle episodic memory lapses, especially for recent events
- Word-finding pauses and slowed language production
- Reduced mental flexibility and planning difficulties
- Visuospatial slips, like getting lost on familiar routes
- Apathy, anxiety, or mood shifts that are new and persistent
- Mild changes in judgment or financial management
These features may emerge gradually and fluctuate, which is why serial assessments are so useful and why collateral history from partners or relatives is so valuable.
Distinguishing look-alikes
Not all cognitive decline is Alzheimer’s, and early clarity matters for care. Dementia with Lewy bodies can cause prominent fluctuations, visual hallucinations, REM sleep behavior disorder, and early parkinsonism. Limbic-predominant age-related TDP-43 encephalopathy (LATE) can mirror amnestic profiles in the oldest-old. Vascular injury—often “silent” microinfarcts—can erode processing speed and executive function. A combination of neuropsychology, MRI, and fluid or imaging biomarkers narrows these differentials and guides tailored management.
Why earlier answers change outcomes
Even where drugs have modest symptomatic benefit, earlier diagnosis still reshapes the trajectory. It enables risk-factor optimization for blood pressure, sleep apnea, diabetes, and hearing loss; initiates cognitive, social, and physical stimulation; and supports planning for safety, driving, and financial decisions. Families gain time to assemble resources, reduce crises, and set realistic, person-centered goals.
Crucially, as disease-modifying approaches advance, the window for maximal benefit is early—before widespread neuronal loss. That makes accurate, timely identification of the first signs not just clinically satisfying but ethically and practically essential.
The path ahead
The future points toward streamlined blood tests, refined imaging protocols, and integrated diagnostic algorithms that combine clinical, cognitive, and biological signals. Equitable access will be a key challenge, as will clear communication that separates risk from diagnosis and disease labels from the person behind the symptoms. With better tools and earlier action, living well with Alzheimer’s becomes a more realistic, shared goal.